Immune-mediated hepatitis with the Moderna vaccine, no longer a coincidence but confirmed
Tun et al., Journal of Hepatology, 2021.10
Discussion: “This case illustrates immune-mediated hepatitis secondary to the Moderna vaccine, which on inadvertent re-exposure led to worsening liver injury with deranged synthetic function. This occurred in a well man with no other medical problems. The onset of jaundice associated with the mRNA vaccine was unusually rapid. This was also illustrated in the other cases where symptoms developed over a median of 7 days (range 4-35).
The mRNA vaccine pathway triggers pro-inflammatory cytokines including interferon and cross-reactivity has been illustrated between the antibodies against the spike protein and self-antigens.
Seven cases of suspected immune-mediated hepatitis have been reported with SARS-2-COV mRNA vaccines (3 with Pfizer and 4 with Moderna). Liver histology was assessed in every case and findings were similar to ours, indicating acute hepatitis with interface hepatitis, lymphoplasmacytic infiltrate and absence of fibrosis.”
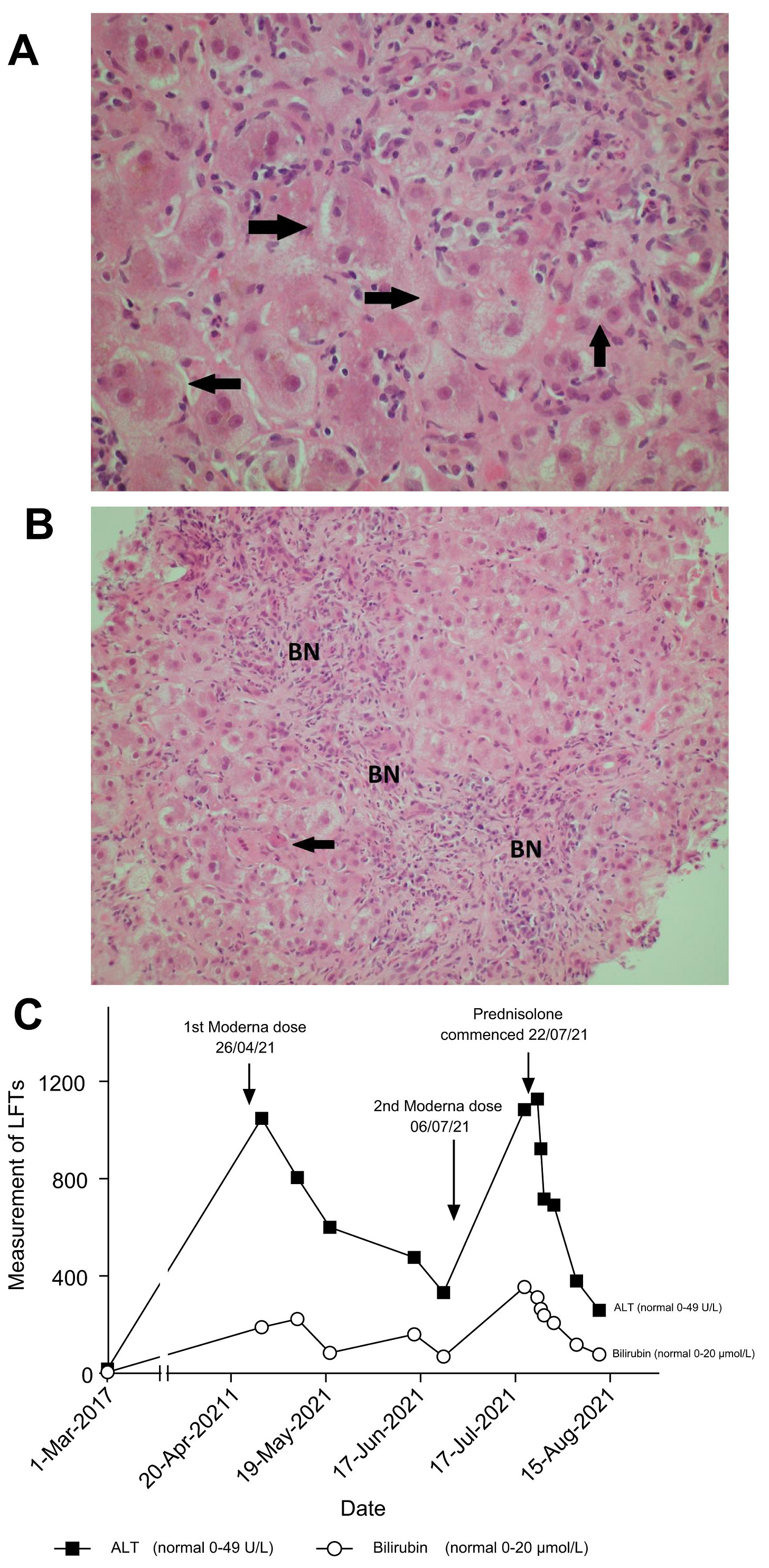
Fig. 1 Histological findings and biochemical findings.
H&E-stained section of liver biopsy indicates acute hepatitis. (A) The parenchymal hepatocytes are arranged into rosette forms (marked with arrows) with cholestasis. (B) BN from hepatocyte loss, some by apoptosis (arrow). (C) Diagram showing trend of bilirubin and ALT following Moderna vaccine dose 1 and 2 with response to prednisolone. ALT, alanine aminotransferase; BN, bridging necrosis.
Understanding Vaccine Causation Conference, WCH, 2022.02.05
How are adverse effects proved? Join moderators Michael Alexander, Dr. Mark Trozzi, Charles Kovess, Megha Verma, and host Shabnam Palesa Mohamed as we unpack this important legal and medical question.
Seneff et al., Authorea, 2022.01.01
“The mRNA SARS-CoV-2 vaccines were brought to market in response to the widely perceived public health crises of Covid-19. The utilization of mRNA vaccines in the context of infectious disease had no precedent, but desperate times seemed to call for desperate measures. The mRNA vaccines utilize genetically modified mRNA encoding spike proteins. These alterations hide the mRNA from cellular defenses, promote a longer biological half-life for the proteins, and provoke higher overall spike protein production. However, both experimental and observational evidence reveals a very different immune response to the vaccines compared to the response to infection with SARS-CoV-2. As we will show, the genetic modifications introduced by the vaccine are likely the source of these differential responses. In this paper, we present the evidence that vaccination, unlike natural infection, induces a profound impairment in type I interferon signaling, which has diverse adverse consequences to human health. We explain the mechanism by which immune cells release into the circulation large quantities of exosomes containing spike protein along with critical microRNAs that induce a signaling response in recipient cells at distant sites. We also identify potential profound disturbances in regulatory control of protein synthesis and cancer surveillance. These disturbances are shown to have a potentially direct causal link to neurodegenerative disease, myocarditis, immune thrombocytopenia, Bell’s palsy, liver disease, impaired adaptive immunity, increased tumorigenesis, and DNA damage. We show evidence from adverse event reports in the VAERS database supporting our hypothesis. We believe a comprehensive risk/benefit assessment of the mRNA vaccines excludes them as positive contributors to public health, even in the context of the Covid-19 pandemic.”
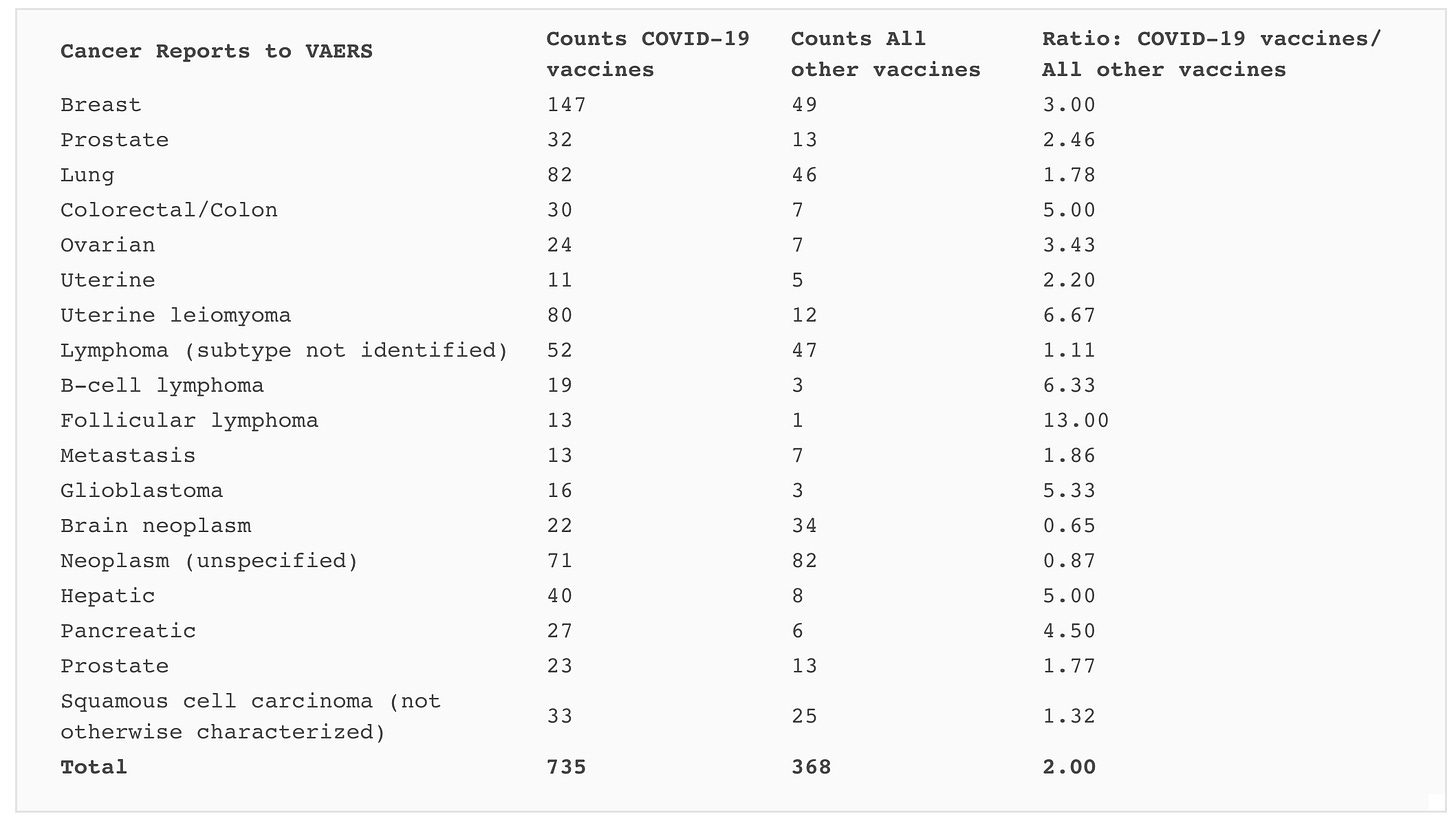
Table 1. Number of events in the VAERS database from 1990 to December 12, 2021, where several terms indicating cancer occurred in association with COVID-19 vaccines or with all other vaccines, along with the ratio between the two counts. Counts were restricted to data from the United States. Note that counts for all the other vaccines are totals for 31 years, whereas the COVID-19 counts are for a single class of vaccines over less than one year.
“COVID-19 Vaccines & neurodegenerative disease”, Dr. S. Seneff, 2022.01.03, 📹
“How COVID shots suppress your immune system”, Dr. S. Seneff, 2022.01, 📹
“Possible unintended consequences of the mRNA vaccines”, Seneff, 2021.07, 📹
Rose, Science, Public Health Policy & the Law, 2021.10
“Following the initiation of the global rollout and administration of the COVID-19 vaccines1,2 on December 17, 2020, in the United States, hundreds of thousands of individuals have reported Adverse Events (AEs) using the Vaccine Adverse Events Reports System (VAERS). To date, approximately 50% of the population of the United States have received 2 doses of the COVID-19 products with 427,831 AEs reported into VAERS as of August 6th, 2021.
Pharmacovigilance (PV) is the process of collecting, monitoring, and evaluating AEs for safety signals to reduce harm to the public in the context of pharmaceutical and biological agents. Many of the issues with VAERS are becoming well known – especially with regards to reporting and recording of data – in light of the extensive use of this system this year, challenging its functionality as a pharmacovigilance system.
This appraisal assesses three issues that respond to the question of VAERS pharmacovigilance by analyzing VAERS data: 1. deleted reports, 2. delayed entry of reports and 3. recoding of Medical Dictionary for Regulatory Activities (MedDRA) terms from severe to mild. The most recently updated publicly available VAERS dataset was found to have N=1516 (0.4%) VAERS IDs removed (“missing”).”
Dr. Jessica Rose, Presentation of VAERS data to the World Council for Health, 2021.10
Dr. Jessica Rose, VAERS update for the Canadian COVID Care Alliance, 2021.09
COVID-19 mRNA vaccination leading to CNS inflammation: a case series
Khayat-Khoei et al., Journal of Neurology, 2021.09
“The availability of vaccines against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), provides hope towards mitigation of the coronavirus disease 2019 (COVID-19) pandemic. Vaccine safety and efficacy has not been established in individuals with chronic autoimmune diseases such as multiple sclerosis (MS). Anecdotal reports suggest that the vaccines may be associated with brain, spinal cord, peripheral nervous system, and cardiac inflammation. Based on the high morbidity and unpredictable course of COVID-19, and the need to achieve herd immunity, vaccination has been recommended for patients with MS. We report clinical and MRI features of seven individuals who received the Moderna (n = 3) or Pfizer (n = 4) SARS-CoV-2 mRNA vaccines. Within one to 21 days of either the first (n = 2) or second (n = 5) vaccine dose, these patients developed neurologic symptoms and MRI findings consistent with active CNS demyelination of the optic nerve, brain, and/or spinal cord. Symptoms included visual loss, dysmetria, gait instability, paresthesias, sphincter disturbance, and limb weakness. Age ranged from 24 to 64 (mean 39.1) years; five were woman (71.4%). The final diagnosis was exacerbation of known stable MS (n = 4, two were receiving disease-modifying therapy at the time of vaccination), new onset MS (n = 2), or new onset neuromyelitis optica (n = 1). All responded to corticosteroid (n = 7) or plasma exchange (n = 1) therapy, with five returning to baseline and two approaching baseline. Large prospective studies are required to further investigate any possible relationship between COVID-19 vaccines and acute CNS demyelination.”
Fig. 5. 3 T MRI in case 5. Brain: axial (a–c) T2 FLAIR obtained 39 days after new neurologic symptom onset, showing periventricular and juxtacortical T2 lesions typical of multiple sclerosis; note one of the lesions ring-enhances with gadolinium (d, white arrow). Spinal cord: thoracic sagittal STIR (e) shows a T2 hyperintense spinal cord lesion suggestive of multiple sclerosis (white arrow). No cervical spinal cord enhancing lesions or new cervical spinal cord T2 lesions were seen (not shown)
Dr. S. Seneff, COVID-19, Glyphosate, Deuterium and SARS-CoV-2 Vaccines, 2021.08
Einstein et al., Cureus, 2021.06
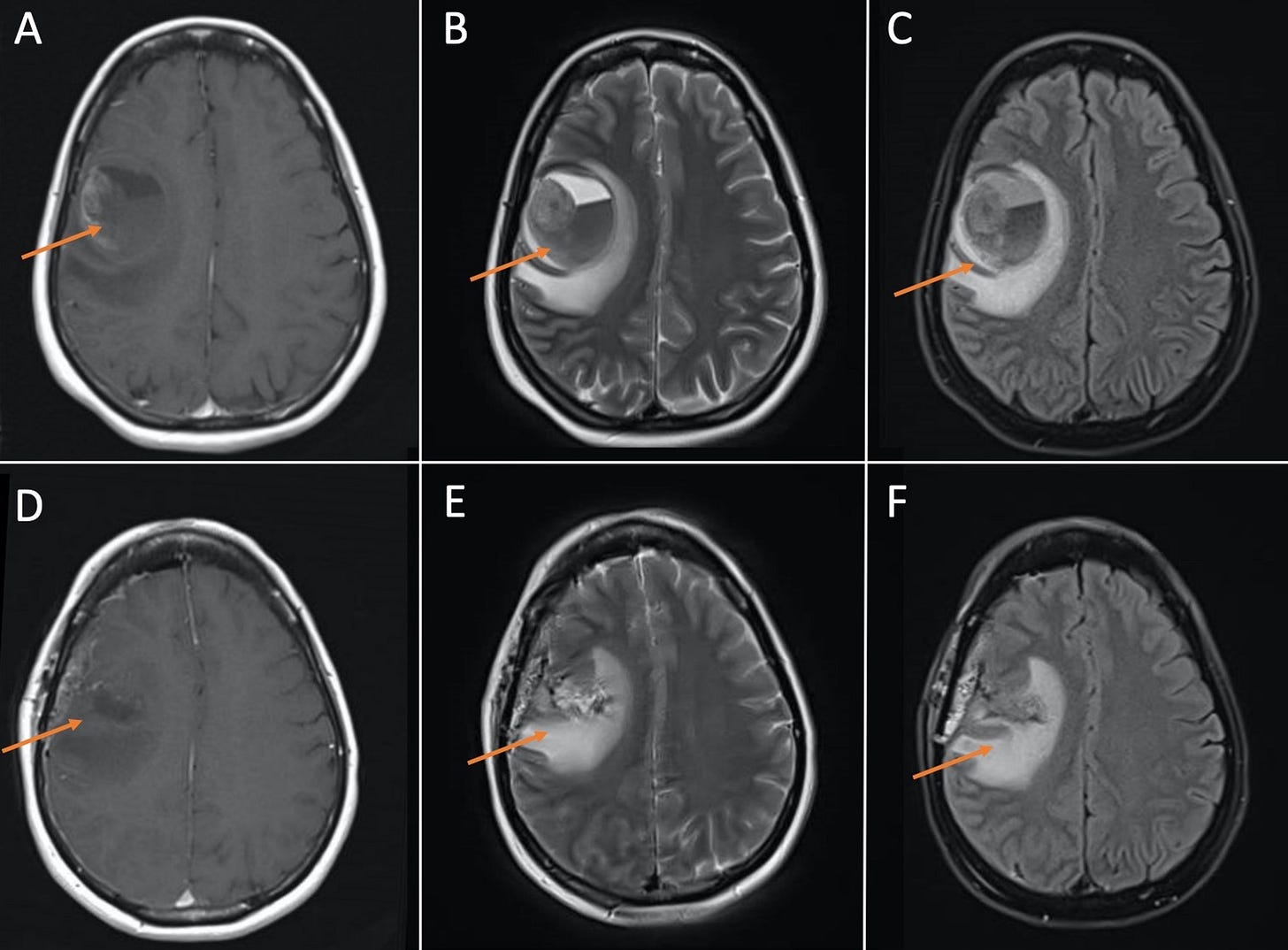
“A global effort is underway to distribute coronavirus disease 2019 (COVID-19) vaccines to limit the crisis. Although adverse events related to vaccination are rare, there have been cases of new-onset neurologic symptoms following vaccination. We present two cases of new-onset neurologic symptoms post-vaccination that, upon further workup, revealed two different neuro-oncologic processes requiring neurosurgical intervention and further treatment. We hypothesize that despite these processes being unrelated to vaccination, the COVID-19 vaccines may induce an inflammatory cascade with the ability to uncover underlying sinister pathology. Our report therefore emphasizes the need for careful evaluation in the setting of new-onset neurologic symptoms after COVID-19 vaccination.”
Figure 1: MRI brain demonstrating large hemorrhagic cavity in the right frontal lobe with an enhancing focus along the right superolateral margin representing a hemorrhagic mass. (A), (B), and (C) are pre-operative T1, T2, and FLAIR images, respectively. (D), (E), and (F) are post-operative T1, T2, and FLAIR images, respectively, demonstrating resection of mass and post-surgical changes. FLAIR: fluid-attenuated inversion recovery.
Seneff and Nigh, Int. Journal of Vaccine Theory, Practice, & Research, 2021.05
“Operation Warp Speed brought to market in the United States two mRNA vaccines, produced by Pfizer and Moderna. Interim data suggested high efficacy for both of these vaccines, which helped legitimize Emergency Use Authorization (EUA) by the FDA. However, the exceptionally rapid movement of these vaccines through controlled trials and into mass deployment raises multiple safety concerns. In this review we first describe the technology underlying these vaccines in detail. We then review both components of and the intended biological response to these vaccines, including production of the spike protein itself, and their potential relationship to a wide range of both acute and long-term induced pathologies, such as blood disorders, neurodegenerative diseases and autoimmune diseases. Among these potential induced pathologies, we discuss the relevance of prion-protein-related amino acid sequences within the spike protein. We also present a brief review of studies supporting the potential for spike protein “shedding”, transmission of the protein from a vaccinated to an unvaccinated person, resulting in symptoms induced in the latter. We finish by addressing a common point of debate, namely, whether or not these vaccines could modify the DNA of those receiving the vaccination. While there are no studies demonstrating definitively that this is happening, we provide a plausible scenario, supported by previously established pathways for transformation and transport of genetic material, whereby injected mRNA could ultimately be incorporated into germ cell DNA for transgenerational transmission. We conclude with our recommendations regarding surveillance that will help to clarify the long-term effects of these experimental drugs and allow us to better assess the true risk/benefit ratio of these novel technologies.”
Figure 2: A simple model for a process by which the spike protein produced through the mRNA vaccines could induce a pathological response distinct from the desirable induction of antibodies to suppress viral entry. Redrawn with permission from Suzuki and Gychka, 2021.
Rose, Science, Public Health Policy & the Law, 2021.05
“Following the global roll-out and administration of the Pfizer/BioNTech (BNT1 62b2) and Moderna (mRNA-1 273) COVID-1 9 vaccines1 on December 1 7, 2020 in the United States, and of the Janssen COVID-1 9 Vaccine PF (produced by Johnson & Johnson) on April 1 st, 2021 , tens of thousands of individuals have reported adverse events (AEs) using the Vaccine Adverse Events Reports System (VAERS). This work summarizes this data to date and serves as information for the public and a reminder of the relevance of any adverse events, including deaths, that occur as a direct result of biologicals as prophylactic treatments. This is especially relevant in the context of technologically novel treatments in the experimental phase of development. Analysis suggests that the vaccines are likely the cause of reported deaths, spontaneous abortions, anaphylactic reactions and cardiovascular, neurological and immunological AEs. The precautionary principle promotes transparency and the adoption of preventative measures to address potential risks to the public in the arena of vaccination programs, and it is vital that individuals are informed of these potential risks before agreeing to participate in any medically involved treatment program. VAERS reporting and recording is essential to the proper functioning of this system. It cannot be over-emphasized that the public should know how to use this system such that they actually do use it, and that once reports are made, responsible individuals enter each report into the database accordingly.”
Kowarz et al., Research Square preprint, 2021.05
“During the last months many countries have started the immunization of millions of people by using vector-based vaccines. Unfortunately, severe side effects became overt during these vaccination campaigns: cerebral venous sinus thromboses (CVST), absolutely rare under normal life conditions, were found as a severe side effect that occured 4-14 days after first vaccinations. Besides CVST, Splanchnic Vein Thrombosis (SVT) was also observed. This type of adverse event has not been observed in the clinical studies of AstraZeneca, and therefore led immediately to a halt in vaccinations in several european countries. These events were mostly associated with thrombocytopenia, and thus, similar to the well-known Heparin-induced thrombocytopenia (HIT). Meanwhile, scientists have proposed a mechanism to explain this vaccine-induced thrombocytopenia. However, they do not provide a satisfactory explanation for the late thromboembolic events. Here, we present data that may explain these severe side effects which have been attributed to adenoviral vaccines. According to our results, transcription of wildtype and codon-optimized Spike open reading frames enables alternative splice events that lead to C-terminal truncated, soluble Spike protein variants. These soluble Spike variants may initiate severe side effects when binding to ACE2-expressing endothelial cells in blood vessels. In analogy to the thromboembolic events caused by Spike protein encoded by the SARS-CoV-2 virus, we termed the underlying disease mechanism the “Vaccine-Induced Covid-19 Mimicry” syndrome (VIC19M syndrome).”
1 A: Splice site prediction within the Spike open reading frames Splice site prediction was carried out by using SpliceRoover. Splice donor sites are given in red, splice acceptor sites in green. SpliceRoover calculates splice sites with a score between 0 to 1, but only splice site with >0,15 were displayed. All sites were displayed in 4 colors as indicated. All splice sites are numbered with “0”, “+1” or “+2”, to indicate how the open reading frame is disrupted. … 1B: Splice trap and Luciferase experiments in HEK293T cells The 3 different splice traps were cloned into the pSBbi-GP vector. This vector encodes two polycistronic transcripts, one encoding GFP and Puromycin resistance (from left to right), while the other encodes the full-length Spike gene fused to an artificial intron and an ATG-deleted Luciferase gene. Splice events to the Luciferase will result in “in-frame” and “out-of-frame” Spike-Luciferase fusion transcripts. … 1C: Ad5-Spike and AZD1222 transduction experiments in Hela cells Two adenoviral constructs were used to infect Hela cells (HAd5::Spike and AZD1222). After RNA isolation specic RT-PCR experiments were performed. The transgene in the HAd5::Spike vector is 99% identical to the AZ and J&J sequence and contains all splice donor sites of the AZ vector, and all splice acceptors of the J&J vector. … 1D: Predicted disease mechanism We predict for the VIC19M syndrome that rare side effects are caused by two independent mechanism: one is the postulated VITT mechanism that is accompanied by the thrombozytopenic situation, while the other mechanism is based on the availability of soluble Spike protein variants in a systemic fashion. We also postulate that soluble Spike protein variants are able to bind to ACE2 expressing endothelial cells which in turn may trigger an ADCC-like mechanisms that result in thromboembolic events (CVST and SVT). Thus, the presence of auto-autobodies against PF4 in combination with soluble Spike protein variants that bind to ACE2 expressing endothelial cells may explain the rare severe side effects (estimated 1 in 120,000 vaccinated persons in Europe).
Smith, Authorea, 2021.03
“BNT162b2 vaccine against Covid-19 is composed of an RNA having 4284 nucleotides, divided into 6 sections, which bring the information to create a factory of S Spike proteins, the ones used by Sars-CoV-2 (Covid-19) to infect the host. After that, these proteins are directed outside the cell, triggering the immune reaction and antibody production.
The problem is the heavy alteration of the mRNA: Uracil is replaced to fool the immune system with Ψ (Pseudouridine); the letters of all codon triplets are replaced by a C or a G, to extremely increase the speed of protein synthesis; replacement of some amino acids with Proline; addition of a sequence (3'-UTR) with unknown alteration.
These impairments could cause strong doubts about the presence of codon usage errors. An eventual mistranslation has consequences on the pathophysiology of a variety of diseases.In addition, mRNA injected is a pre-mRNA, which can lead to the multiple mature mRNAs; these are alternative splicing anomalies, direct source of serious long-term harm on the human health.
In essence, what will be created may not be identical with protein S Spike: just an error in translational decoding, codons misreading, production of different amino acids, then proteins, to cause serious long-term damage to human health, despite the DNA is not modified, being instead in the cell nucleus and not in the cytoplasm, where the modified mRNA arrives.
However, in this case, the correlation between speed of synthesis and protein expression with synthesis errors, as well as the mechanism that could affect the translation of the sequence remain obscure, many trials have not yet been performed.”