Autoimmunity: From Bench to Bedside, Anaya et al., 2013 (pdf)
An introduction to immunology and immunopathology (Marshall et al., 2018)
ACE2-independent infection of T lymphocytes by SARS-CoV-2
Shen et al., Signal Transduction and Targeted Therapy, 2022.03
“SARS-CoV-2 induced marked lymphopenia in severe patients with COVID-19. However, whether lymphocytes are targets of viral infection is yet to be determined, although SARS-CoV-2 RNA or antigen has been identified in T cells from patients. Here, we confirmed that SARS-CoV-2 viral antigen could be detected in patient peripheral blood cells (PBCs) or postmortem lung T cells, and the infectious virus could also be detected from viral antigen-positive PBCs. We next prove that SARS-CoV-2 infects T lymphocytes, preferably activated CD4 + T cells in vitro. Upon infection, viral RNA, subgenomic RNA, viral protein or viral particle can be detected in the T cells. Furthermore, we show that the infection is spike-ACE2/TMPRSS2-independent through using ACE2 knockdown or receptor blocking experiments. Next, we demonstrate that viral antigen-positive T cells from patient undergone pronounced apoptosis. In vitro infection of T cells induced cell death that is likely in mitochondria ROS-HIF-1a-dependent pathways. Finally, we demonstrated that LFA-1, the protein exclusively expresses in multiple leukocytes, is more likely the entry molecule that mediated SARS-CoV-2 infection in T cells, compared to a list of other known receptors. Collectively, this work confirmed a SARS-CoV-2 infection of T cells, in a spike-ACE2-independent manner, which shed novel insights into the underlying mechanisms of SARS-CoV-2-induced lymphopenia in COVID-19 patients.”
Fig. 1 Peripheral blood lymphocytes are infected by SARS-CoV-2 in COVID-19 patients. a Percentage of different types of lymphocytes in the healthy donors (n = 15) or in COVID-19 patients (n = 22). (b) Percentage of CD4 + and CD8 + T lymphocytes in healthy donor (n = 8) or in COVID-19 patients (n = 9). c, d Immunofluorescent test of the presence of SARS-CoV-2 viral antigen in T cells. PBCs (c) or postmortem lung section (d) from COVID-19 patients were stained with T lymphocytes (CD3, green), SARS-CoV-2 (NP, red) and nuclei (DAPI, blue). In-house-made pAb against SARS-CoV-2 NP was used. White arrows indicate areas T lymphocytes that were infected by SARS-CoV-2. Pictures were taken under confocal microscopy with a bar = 50 μm (c) or 200 μm (d). Comparison of mean values (a, b) between two groups was analyzed by Student’s t test. *P < 0.05; **P < 0.01; ****P < 0.0001; NS no significance
Abramczyk et al., bioRxiv, 2022.03
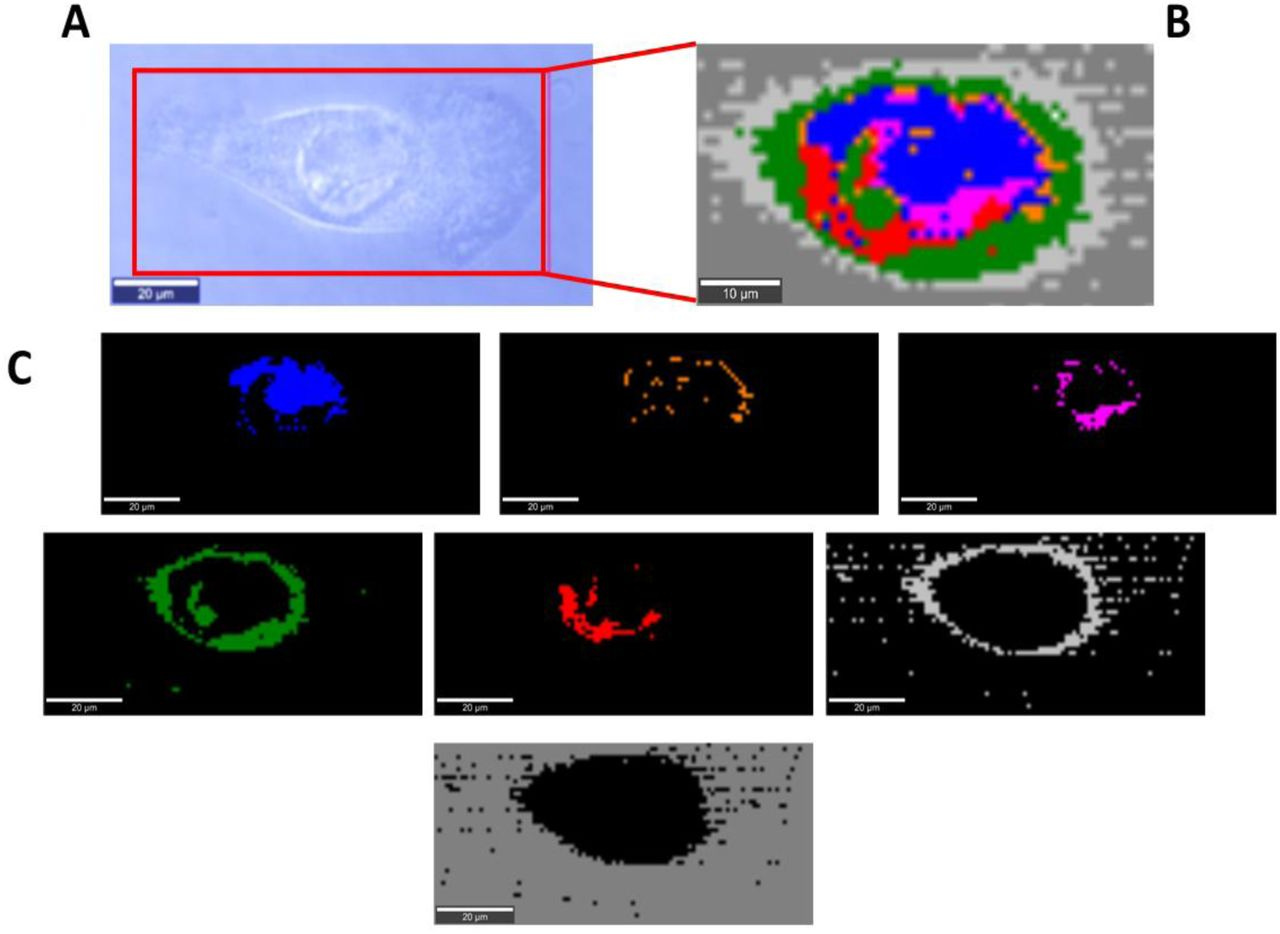
The paper presents the effect of COVID-19 mRNA (Pfizer/BioNT) vaccine on in vitro glial cells of the brain studied by means of Raman spectroscopy and imaging.. The results obtained for human brain normal and tumor glial cells of astrocytes, astrocytoma, glioblastoma incubated with the Covid-19 mRNA vaccine Pfizer/BioNT vaccine show alterations in the reduction-oxidation pathways associated with Cytochrome c.
We found that the Pfizer/BioNT vaccine down regulate the concentration of cytochrome c in mitochondria upon incubation with normal and tumorous glial cells. Concentration of oxidized form of cytochrome c in brain cells has been shown to decrease upon incubation the mRNA vaccine. Lower concentration of oxidized cytochrome c results in lower effectiveness of oxidative phosphorylation (respiration), reduced apoptosis and lessened ATP production. Alteration of Amide I concentration, which may reflect the decrease of mRNA adenine nucleotide translocator. Moreover, mRNA vaccine leads to alterations in biochemical composition of lipids that suggest the increasing role of signaling. mRNA vaccine produce statistically significant changes in cell nucleus due to histone alterations. The results obtained for mitochondria, lipid droplets, cytoplasm may suggest that COVID-19 mRNA (Pfizer/BioNT) vaccine reprograms immune responses. The observed alterations in biochemical profiles upon incubation with COVID-19 mRNA in the specific organelles of the glial cells are similar to those we observe for brain cancer vs grade of aggressiveness.”
Figure 3: Microscopy image (A), Raman image of astrocyte (NHA) cell (100×45 µm, resolution 1.0 µm) incubated with Pfizer/BioNT vaccine (dose 60 µL/mL) for 96 hours (B) and Raman imagines of specific organelles: lipids and lipid droplets (blue and orange), mitochondria (magenta),nucleus (red), cytoplasm (green), membrane (light grey), cell environment (dark grey) at 532 nm.
Röltgen et al., Cell, 2022.01
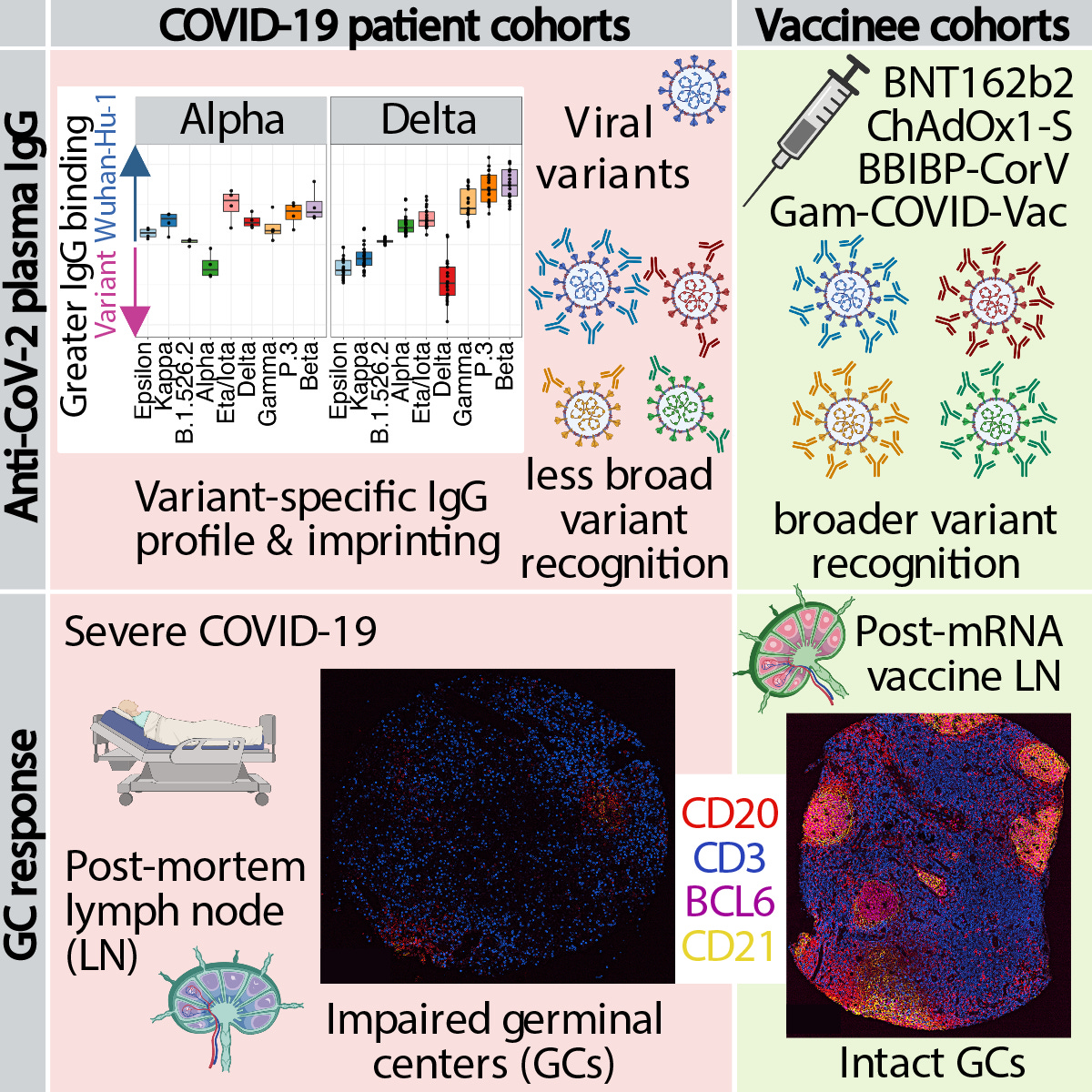
“Vaccination confers broader IgG binding of variant RBDs than SARS-CoV-2 infection
Imprinting from initial antigen exposures alters IgG responses to viral variants
Histology of mRNA vaccinee lymph nodes shows abundant germinal centers
Vaccine spike antigen and mRNA persist for weeks in lymph node germinal centers”
“During the SARS-CoV-2 pandemic, novel and traditional vaccine strategies have been deployed globally. We investigated whether antibodies stimulated by mRNA vaccination (BNT162b2), including 3rd dose boosting, differ from those generated by infection or adenoviral (ChAdOx1-S and Gam-COVID-Vac) or inactivated viral (BBIBP-CorV) vaccines. We analyzed human lymph nodes after infection or mRNA vaccination for correlates of serological differences. Antibody breadth against viral variants is less after infection compared to all vaccines evaluated, but improves over several months. Viral variant infection elicits variant-specific antibodies, but prior mRNA vaccination imprints serological responses toward Wuhan-Hu-1 rather than variant antigens. In contrast to disrupted germinal centers (GCs) in lymph nodes during infection, mRNA vaccination stimulates robust GCs containing vaccine mRNA and spike antigen up to 8 weeks post-vaccination in some cases. SARS-CoV-2 antibody specificity, breadth and maturation are affected by imprinting from exposure history, and distinct histological and antigenic contexts in infection compared to vaccination.”
Geert Vanden Bossche, DVM, PhD, Voice For Science and Solidarity, 2021.12
Conclusion
“The vast majority of medical doctors have very poor knowledge of immunology (let alone vaccinology), which isn’t a part of their curriculum. This is, of course, problematic as they are the critical point of care and advice to patients. It is striking how specialization in one or another medical subdiscipline leads some specialized doctors to have tunnel vision in that they fail to explore cross-disciplinary aspects, even when it comes to solving health challenges, they are not familiar with and are inherently complex and multifaceted. It is, indeed, mind blowing and highly worrisome that - although sailing uncharted waters - some pediatricians are not afraid of blindly criticizing knowledgeable experts and posting their poorly informed viewpoints on the internet, not realizing that the narrative they support in favor of C- 19 vaccination of children implies a formidable individual and public health risk. As extensively documented on my website (www.voiceforscienceandsolidarity.org), I’ve repeatedly debunked ‘immunologically naïve’ comments made by a number of such ‘know-it-all’ doctors. A majority lack the multidisciplinary background required to put the pieces of this complex puzzle together. I’ve, therefore, written an article that should help people to navigate through the heavily polluted opinion landscape while doing their own research: “Some guidance to separating the wheat from the chaff”. I can only recommend these ‘know-it-all’ doctors to first gain some better understanding of some fundamental principles in immunology and read some scientific peer- reviewed publications referred to on our website before trying to cross the boundaries of their knowledge.
Innate immunity is the holy grail for protection against productive infection and hence, the key to sterilizing and herd immunity. Vaccinal, S-directed Abs of high affinity prevent binding of multi-specific innate Abs that have low affinity for S; natural booster immunizations due to dominant circulation of highly infectious variants are likely to sustain this vaccine-induced immune suppression as do booster jabs.
Children represent the population group with the most abundant innate immune capacity; erosion of their innate immune defense makes them vulnerable, not only to Covid-19 disease (as C-19 vaccines protect at best against severe disease) but also to disease caused by other respiratory viruses; in addition, suppression of relevant innate Abs is likely to lead to an increased incidence of autoimmune disease (see above).
Emerging evidence reveals that the unvaccinated may be benefiting from ‘training’ their innate immune system through exposure to the virus. This suggests that an ever-increasing part of the unvaccinated population may enjoy improved protection from infection, and certainly from disease, as time goes by (an objective criterion that will be easy to verify!).
Indeed, one can only hope that medical doctors who are currently pushing the C-19 mass vaccination agenda will abandon their silo thinking and educate themselves on immunology and vaccinology to gain a much better understanding of the catastrophic consequences continued mass vaccination will lead to, not least of which for children. Given the Hippocratic Oath they have taken, that’s the least one can expect from medical experts who feel called to make recommendations on mass vaccination of children.”
Wong and Perlman, Nature Reviews Immunology, 2021.11
“Analyses of patients have revealed marked dysregulation of the immune system in severe cases of human coronavirus infection, and there is ample evidence that aberrant immune responses to human coronaviruses are typified by impaired induction of interferons, exuberant inflammatory responses and delayed adaptive immune responses. In addition, various viral proteins have been shown to impair interferon induction and signalling and to induce inflammasome activation. This suggests that severe disease associated with human coronaviruses is mediated by both dysregulated host immune responses and active viral interference. Here we discuss our current understanding of the mechanisms involved in each of these scenarios.”
Fig. 2. Coronavirus RNA in the cytoplasm is sensed by the cytoplasmic RNA sensors RIG-I and MDA5. Sensing of viral RNA triggers conformational changes in these sensors and results in the recruitment of downstream effector proteins. … Viral proteins of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and related coronaviruses shown in the figure interfere with interferon production and signalling at different steps. Viral proteins (depicted in red) are from SARS-CoV-2 unless otherwise specified.
5+1 Concerns about SARS-CoV2 Biology: A Call to Pause, Deliberate and Revise Policy
Couey and Stover, 2021.10
What is non-sterilizing immunity and why do asymptomatic infections in immunized populations increase the risks of the current non-sterilizing immunization campaigns?
Why does the SARS-CoV2 origin and unique “spike” protein biology matter to developing effective pandemic solutions?
How does the SARS-CoV2’s binding strength for ACE2 protein relate to antibody dependent enhancement (ADE) in humans and why does this matter to immunization policy?
Why could this first EUA immunization present a risk to a child’s developing immune system?
What are “viral swarms” and why do they impact collective (herd) immunity and a path to end this pandemic?
Why the current suite of “investigational vaccines” should be considered “transfections” or “transformations” rather than vaccines?
Conclusion: “In the challenge to make decisions and adjust direction in time of crisis, the global community is still learning about the virus and the ramifications of human intervention. In this environment, access to information, analysis, and approaches are paramount. Decisions today have long term consequences for global health. While it is tempting to look to EUA immunizations as a single foolproof solution, there are personal and broader risks that are not yet well appreciated. Moreover, a multilayered approach that considers the full range of tools and options available will ensure greater resilience and success beyond 2021.”
Sterilizing versus non-sterilizing immunity. A. Sterilizing immunity developed after vaccination against measles virus. Multiple targets (epitopes) are the basis for lifelong immunity. B. Natural non-sterilizing immunity to coronaviruses and EUA immunization to SARS-CoV2 spike protein are shown. Natural immunity to SARS-CoV2 recognizes targets (blue circles) other than the spike protein (red triangles). Immunization to the spike protein alone selects for variants from future infections that can escape spike protein antibodies. Because spike protein biology is key to severe COVID-19, enriching for spike protein variants could be potentially catastrophic.
The BNT162b2 mRNA vaccine against SARS-CoV-2 reprograms both adaptive and innate immune responses
Föhse et al., medRxiv, 2021.05
“The mRNA-based BNT162b2 vaccine from Pfizer/BioNTech was the first registered COVID-19 vaccine and has been shown to be up to 95% effective in preventing SARS-CoV-2 infections. Little is known about the broad effects of the new class of mRNA vaccines, especially whether they have combined effects on innate and adaptive immune responses. Here we confirmed that BNT162b2 vaccination of healthy individuals induced effective humoral and cellular immunity against several SARS-CoV-2 variants. Interestingly, however, the BNT162b2 vaccine also modulated the production of inflammatory cytokines by innate immune cells upon stimulation with both specific (SARS-CoV-2) and non-specific (viral, fungal and bacterial) stimuli. The response of innate immune cells to TLR4 and TLR7/8 ligands was lower after BNT162b2 vaccination, while fungi-induced cytokine responses were stronger. In conclusion, the mRNA BNT162b2 vaccine induces complex functional reprogramming of innate immune responses, which should be considered in the development and use of this new class of vaccines.”
Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19
Sekine et al., Cell, 2020.08
Highlights
Acute-phase SARS-CoV-2-specific T cells display an activated cytotoxic phenotype
Convalescent-phase SARS-CoV-2-specific T cells generate broad responses
Polyfunctional SARS-CoV-2-specific T cells also occur in seronegative individuals
Summary: “SARS-CoV-2-specific memory T cells will likely prove critical for long-term immune protection against COVID-19. Here, we systematically mapped the functional and phenotypic landscape of SARS-CoV-2-specific T cell responses in unexposed individuals, exposed family members, and individuals with acute or convalescent COVID-19. Acute-phase SARS-CoV-2-specific T cells displayed a highly activated cytotoxic phenotype that correlated with various clinical markers of disease severity, whereas convalescent-phase SARS-CoV-2-specific T cells were polyfunctional and displayed a stem-like memory phenotype. Importantly, SARS-CoV-2-specific T cells were detectable in antibody-seronegative exposed family members and convalescent individuals with a history of asymptomatic and mild COVID-19. Our collective dataset shows that SARS-CoV-2 elicits broadly directed and functionally replete memory T cell responses, suggesting that natural exposure or infection may prevent recurrent episodes of severe COVID-19.”