Park et al., Vaccines, 2022.05
“Cerebral venous thrombosis (CVT), a rare thrombotic event that can cause serious neurologic deficits, has been reported after some ChAdOx1 nCoV-19 vaccinations against coronavirus disease 2019 (COVID-19). However, there are few reports of associations between COVID-19 mRNA vaccination and CVT. We retrospectively analyzed CVT occurrence, time of onset after vaccination, outcomes (recovered/not recovered), and death after COVID-19 vaccination from adverse drug reactions (ADR) reports in VigiBase. A disproportionality analysis was performed regarding COVID-19 mRNA vaccines (BNT162b2 and mRNA-1273) and the ChAdOx1 nCoV-19 vaccine. We identified 756 (0.07%) CVT cases (620 (0.05%) after BNT162b2 and 136 (0.01%) after mRNA-1273) of 1,154,023 mRNA vaccine-related ADRs. Significant positive safety signals were noted for COVID-19 mRNA vaccines (95% lower end of information component = 1.56; reporting odds ratio with 95% confidence interval (CI) = 3.27). The median days to CVT onset differed significantly between the BNT162b2 and ChAdOx1 nCoV-19 vaccines (12 (interquartile range, 3–22) and 11 (interquartile range, 7–16), respectively; p = 0.02). Fewer CVT patients died after receiving mRNA vaccines than after receiving the ChAdOx1 nCoV-19 vaccine (odds ratio, 0.32; 95% CI, 0.22–0.45; p < 0.001). We noted a potential safety signal for CVT occurrence after COVID-19 mRNA vaccination. Therefore, awareness about the risk of CVT, even after COVID-19 mRNA vaccination, is necessary.”
Figure 1. Differences between times of occurrence of cerebral venous thrombosis within 28 days after vaccine administration for each COVID-19 vaccine type. The box plots indicate median numbers of cerebral venous thrombosis cases and interquartile ranges per week depending on each vaccine. There are significant differences in time to cerebral venous sinus thrombosis onset from date of vaccination between the different vaccines within 28 days. In pairwise comparisons between vaccine groups, only significant differences between BNT162b2 and ChAdOx1 nCoV-19 vaccines remained (p = 0.02).
COVID-19 and the Brain: The Neuropathological Italian Experience on 33 Adult Autopsies
Fabbri et al., Biomolecules, 2022.04
“Discussion: From a clinical point of view, different central acute neurological manifestations in patients with COVID-19 have been reported. Ischemic stroke, sinus venous thrombosis and cerebral hemorrhage have been described and have been related to the inflammation, hypoxia, immobilization or diffuse intravascular coagulation. Our findings and the other neuropathological data in the literature seem to confirm this hypothesis. Dizziness, impaired consciousness, confusion, headache, syncope and seizures have also been reported in SARS-CoV-2 infected patients: they are extremely non-specific and could be the manifestation of an ischemic event, such as global cerebral hypoxia, inflammatory status or electrolyte imbalances rather than a direct viral damage of circumscribed cerebral areas.
All these histological data indicate that CNS is probably “a victim more than a target” in SARS-CoV-2: intravascular microthrombi and microinfarcts are in keeping with the hypercoagulable state of severe SARS-CoV-2 infected patients whereas the hypoxic-ischemic general condition, related to the respiratory failure, may indeed be worsened by the consequent brainstem damage appearing as a final event."
Clinical and radiological data suggest that SARS-CoV2 can cause meningitis and encephalitis.”
“Conclusions: The data here shown are consistent with data presently published and features indicate that:
SARS-CoV-2 can directly infect the CNS even if it is not clear which is the direct type of brain tissue damage.
Morphological features indicate that SARS-CoV-2 related coagulation disorders are the main cause of the brain tissue damage. The immediate use of anticoagulant therapy in critical patients reduced thromboembolic complications.
Bacterial superinfection can be the cause of acute leptomeningitis during SARS-CoV-2 infection.
Brain damage can be present even in absence of specific neurological symptoms. Therefore, it is possible that brain involvement could be an underestimated feature in SARS-CoV-2 infected patients.
Astrogliosis, microglial activation and chronic inflammation could play a role for the development of chronic neurological disorders.”
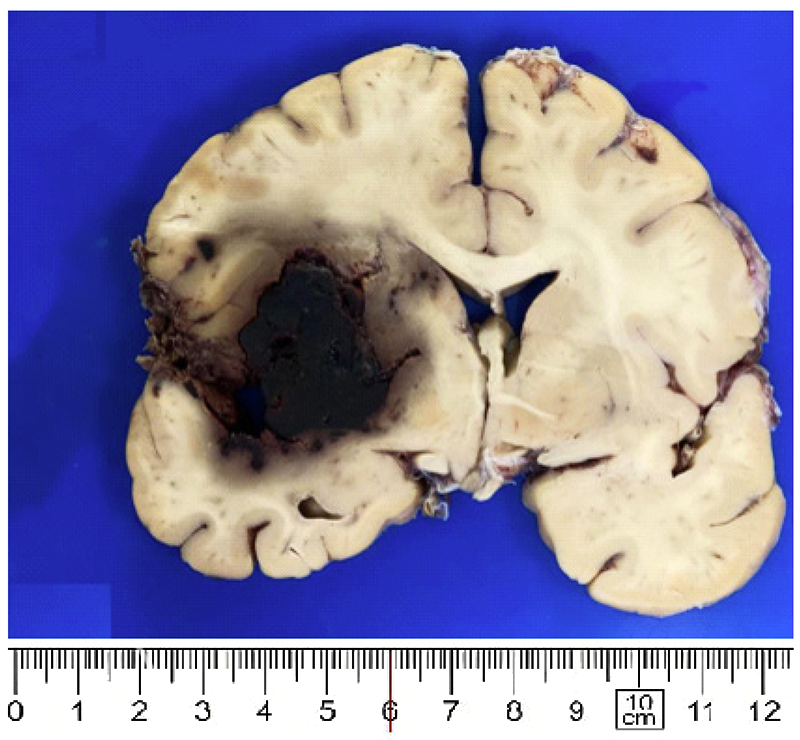
Figure 1. Large hemorrhage in basal ganglia (case 23).
Pomara et al., Pathology – Research and Practice, 2022.03
“This case report describes a fatal case of a young woman with superior sagittal, transverse and sigmoid sinus thrombosis after administration of the ChAdOx1 nCov-19 vaccination. Eleven days post-vaccination she was found unconscious and transferred to the Emergency Department. Blood parameters showed low platelets, and a CT scan showed an extensive left intracranial hemorrhage and the presence of an occlusive thrombus of the superior sagittal sinus. She under-went a craniectomy, but after the intervention, she remained in a comatose state. After a few days, her clinical conditions worsened, and she died. A complete autopsy was performed which showed a thrombosis of the cerebral venous district, of the upper and lower limbs. A blood sample was also performed to carry out a gene study about the predisposition to thrombosis. The organ samples were studied through light microscope both in hematoxylin-eosin and immunohistochemical examination, and showed a strong inflammatory response in all samples and at the site of thrombosis. Our study aims to provide a proper autopsy technique to study the entire cerebral venous system through a multidisciplinary approach (anatomical dissection and neurosurgery) in post-vaccine venous thrombosis.”
Branch retinal vein occlusion in a healthy young man following mRNA COVID-19 vaccination
Pur et al., American Journal of Ophthalmology Case Reports, 2022.02
Purpose: “To report a case of a branch retinal vein occlusion (BRVO) following mRNA COVID-19 vaccination.”
Observations: “A 34-year-old healthy male presented with blurriness in the inferior visual field, intermittent photopsia, multiple retinal hemorrhages, dilated and tortuous retinal vessels, and cotton wools spots in the right eye. The clinical examination and ancillary tests confirmed the diagnosis of a right eye BRVO. The visual symptoms started 2 days following first dose COVID-19 vaccination with the BNT162b2 (Pfizer-BioNTech) mRNA vaccine.”
Conclusions and importance: “This is a rare case of BRVO in an otherwise healthy young man, presenting after vaccination for COVID-19 in the absence of other coagulable risk factors. As the literature on venous thrombosis after COVID-19 vaccinations remains sparse, it is critical to raise awareness that BRVO could be a vaccine-related thrombotic adverse event. We highlight that as more of the population is vaccinated, an increased incidence of BRVO may confirm the link to COVID-19 vaccination.”
“In summary, we report a case of an otherwise healthy young man, presenting with BRVO following a first dose of COVID-19 vaccine in the absence of other coagulable risk factors. Although a direct causal relationship cannot be established, considering the reports of venous thrombosis after COVID-19 vaccination, this finding is suspicious for a vaccine-related thrombotic adverse event. The incidence of RVO either after infection or vaccination appears to be very low, but should prompt ophthalmic examination in patients with even mild visual complaints in these settings. As more of the population is vaccinated, an increased incidence of RVO may confirm the link to COVID-19 vaccination.”
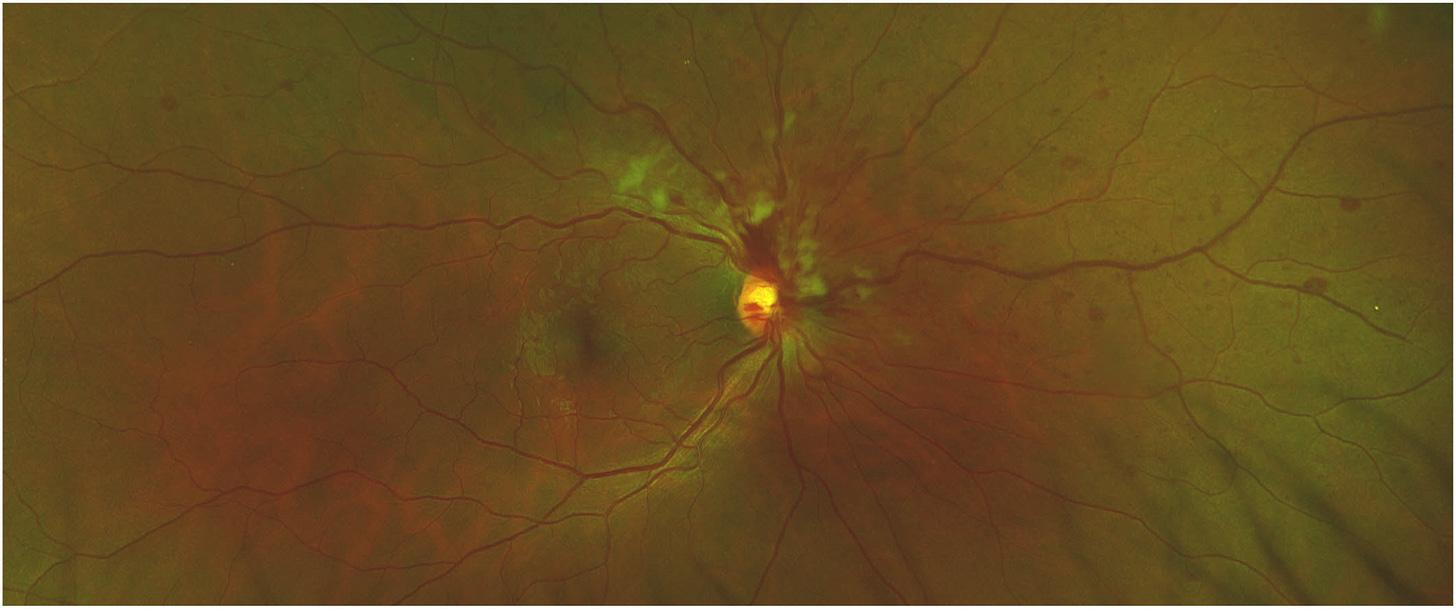
Fig. 1. Colour photo of right eye superior nasal branch retinal vein occlusion demonstrating venous dilation and tortuosity, multiple retinal hemorrhages and cotton wool spots. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
Ischemic Stroke following COVID-19 in a Patient without Comorbidities
Pinzon et al., Case Reports in Medicine, 2021.11
Background. “Stroke is a rare complication of COVID-19. Post-COVID-19 stroke occurs mainly in older patients who have preexisting vascular risk factors. Most strokes are possibly related to hypercoagulability associated with COVID-19 where elevated D-dimer levels were the most common finding. In this case, post-COVID-19 ischemic stroke occurred in a relatively young patient without preexisting cerebrovascular risk factors which were rarely reported before. Case Presentation. A 40-year-old male presented lack of concentration, sluggish mind, and forgetfulness. The patient has a positive COVID-19 history 5 weeks ago. The noncontrast MSCT scan confirmed multifocal lacunar cerebral infarction on the left lateral ventricle. Laboratory tests showed an increase in D-dimer of 1.22 g/ml. Conclusion. In COVID-19 patients without comorbidities, ischemic stroke should be considered.”
Bayas et al., The Lancet, 2021.04
“A 55-year-old woman presented with conjunctival congestion, retro-orbital pain, and diplopia. She had received her first vaccine against SARS-CoV-2— ChAdOx1 nCoV-19—10 days before admission. Both on the night after the vaccination and 7 days later, the patientreportedmarkedflu-likesymptomsandafever. She had no medical history of visual problems, autoimmune disorders, stroke, thrombosis, thrombo- cytopenia, neurological disorders, or arterial disease risk factors—including hypertension, diabetes, or smoking.
That 8, 10, and 18 days after the ChAdOx1 nCoV-19 vaccination, our previously healthy patient developed marked flu-like symptoms, two rare disorders—namely, bilateral SOVT and ITP, and an ischaemic stroke, may indicate a causal relationship. According to the European Medicines Agency review, March 18, health-care professionals should be on the alert for possible cases of thromboembolism—like cerebral venous sinus thrombosis, pulmonary embolus, and deep vein thrombosis—occurring in people who have recently received the ChAdOx1 nCoV-19 vaccine. The thrombotic events—including bilateral SOVT as seen in our patient—may occur in the context of thrombocytopenia”
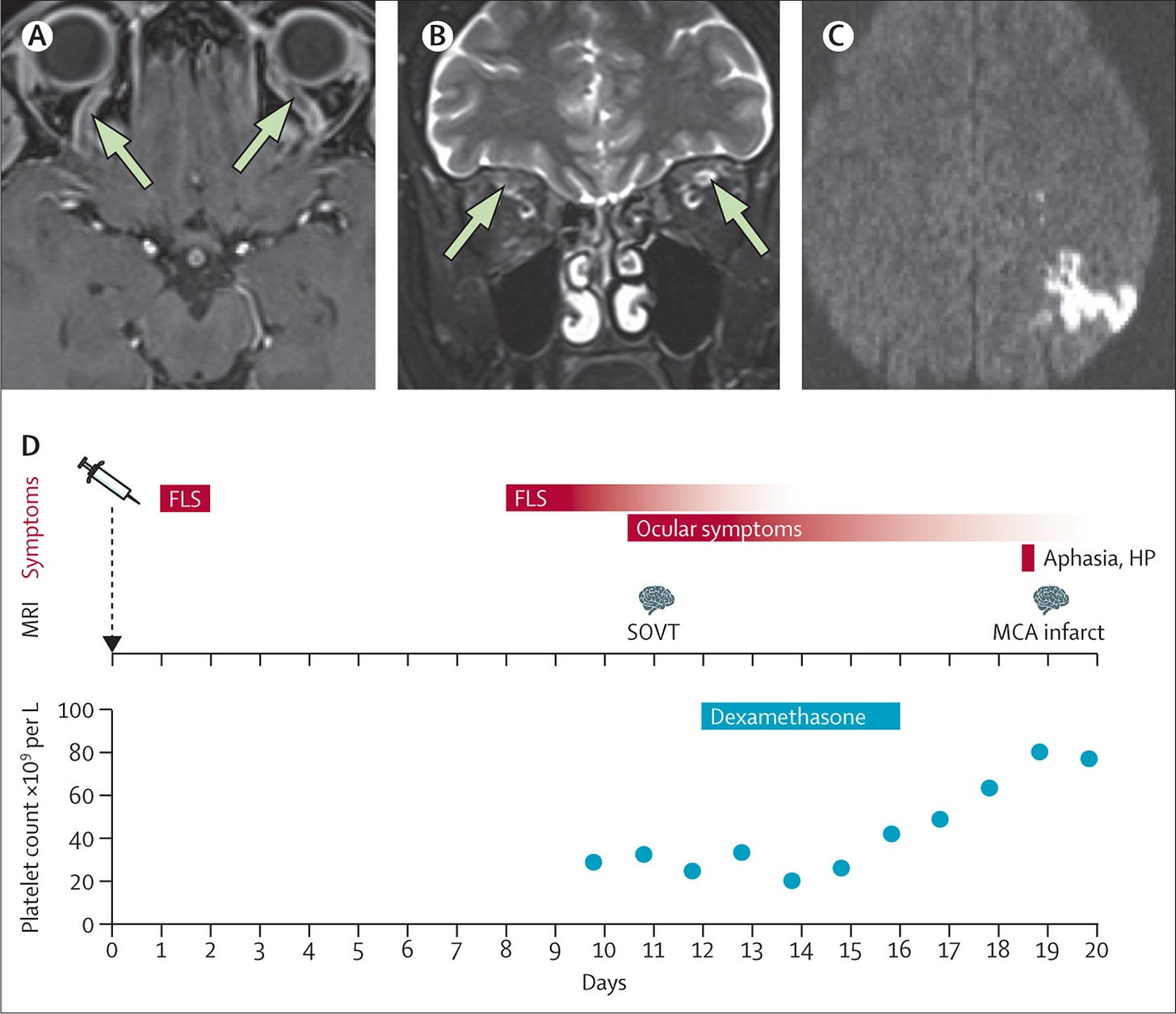
Figure: SOVT and ischaemic stroke after ChAdOx1 nCoV-19 vaccination
MRI shows SOVT with no contrast filling (arrows; A) and bilateral high T2 signal intensity of the superior ophthalmic vein (arrows; B). MRI shows an ischaemic stroke in the left parietal lobe, MCA territory, with restricted diffusion (C). Diagram shows the timeline of symptoms, MRI findings, dexamethasone treatment, and platelet count (D). FLS=flu-like symptoms. HP=hemiparesis. SOVT=superior ophthalmic vein thrombosis. MCA=middle cerebral artery.